AI-Powered Pharmacovigilance Platform
Transform safety case management with automated workflows, real-time analytics, and intelligent insights — all in one unified platform.
Transform safety case management with automated workflows, real-time analytics, and intelligent insights — all in one unified platform.

From case intake to reporting — manage your entire safety workflow in one place
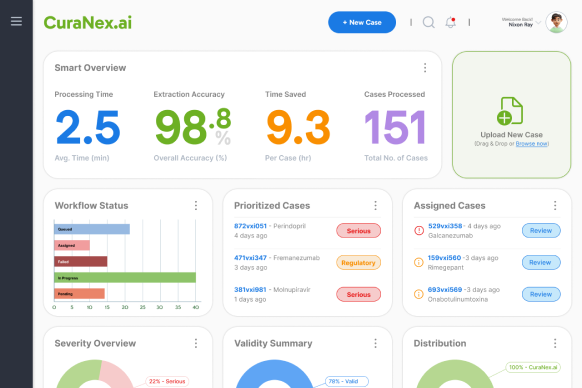
Monitor workflow status, prioritized cases, severity overview, and validity summaries at a glance. Complete visibility into your pharmacovigilance operations.
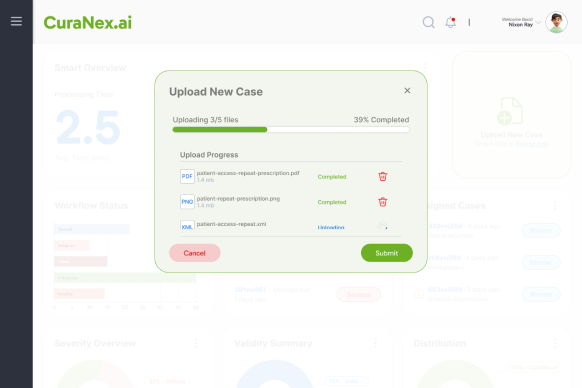
Drag & drop XML files with real-time progress tracking. Automatic validation and error detection ensures data integrity from the start.
Everything you need to manage pharmacovigilance efficiently
Upload, validate, and normalize case data automatically — preserving integrity at every step with intelligent error handling.
Find cases instantly with powerful full-text, faceted, and semantic search across all data fields and documents.
Configure pipelines, escalations, and assignments to match your SOPs perfectly with role-based access control.
Interactive dashboards with severity tracking, validity summaries, and distribution analysis for data-driven decisions.
98.8% extraction accuracy with automated validation, duplicate detection, and comprehensive audit trails.
Natural language queries, smart recommendations, and automated report generation powered by advanced AI.
Bank-level encryption, SOC 2 Type II certified infrastructure, and comprehensive access controls for maximum data protection.
Fully compliant with ISO 27001, SOC 2, HIPAA, GxP, GAMP 3, and GDPR regulations for worldwide operations.
Seamless API connections with existing safety databases, EHR systems, and regulatory reporting platforms.
Explore our intuitive interface designed for safety professionals
Powerful table views with sortable columns, advanced filters, customizable views, and bulk actions. Export to Excel, share with team members, and maintain complete audit trails.
Join safety teams worldwide who trust CuraNex.ai to accelerate reviews, ensure compliance, and surface insights instantly.
See how CuraNex.ai can transform your pharmacovigilance workflow